Access to high-cost innovative medicines in Türkiye
What are the access and pricing challenges facing Pharma in this important developing market? What solutions are available to overcome these challenges?
Merhaba, günaydın, hoşgeldiniz! Today I’m taking you to the beautiful Türkiye. The home of former empires, çay, hot air balloon rides over fairy chimneys, more çay, and crystal clear azure waters.
But you’re here for the MA&P part. So let’s get down to business and explore some of the key factors affecting patient access to high-cost innovative treatments in Türkiye and potential solutions Pharma can use to attain access for them.
The Turkish currency and debt crisis
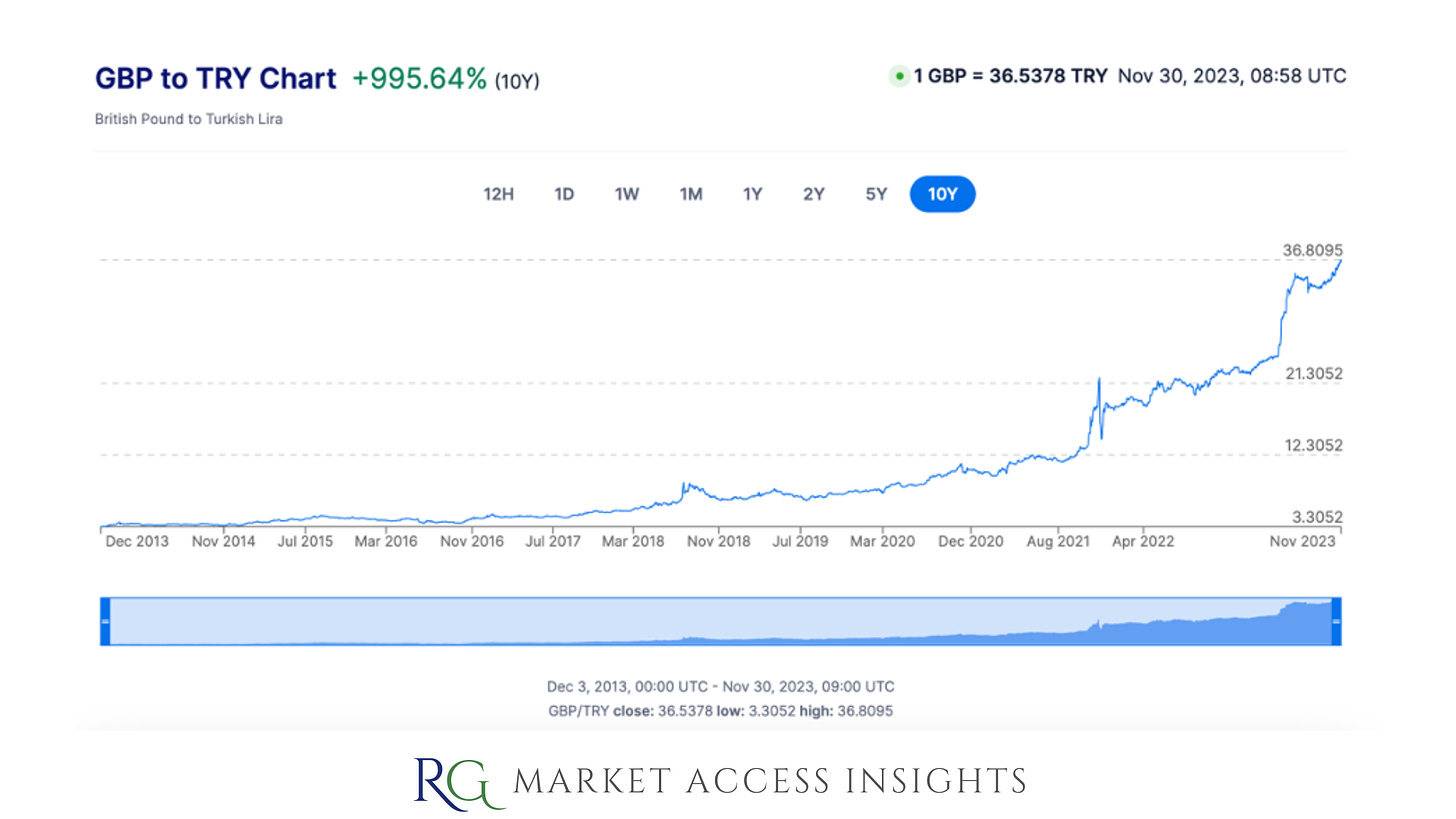
Türkiye has an ongoing currency and debt crisis; the Turkish lira has been plunging in value since 2018, inflation rates are high, borrowing costs are increasing, and loan defaults are becoming more prevalent.
The crisis was caused by the economy's excessive current account deficit, large amounts of private foreign-currency-denominated debt, reduced foreign currency inflows, and “unorthodox” ideas about interest rate policy.
The weakened lira has a significant impact on innovative therapy pricing calculations as we’ll see below. This in turn has impacted access to medicine as some manufacturers are not willing to accept reimbursement prices determined for products covered by the Social Security Institution (SSI).
Additionally, the weakened lira has affected the domestic pharmaceutical industry in Türkiye as local manufacturers rely on imported active pharmaceutical ingredients and export revenues, both of which are highly sensitive to currency exchange rates.
Source for historic exchange rate data.
SSI reimbursed medicines pricing methodology
The reimbursed price of new medicines marketed by foreign manufacturers is determined by the Social Security Institution via a multi-step process, which involves these three key steps:
Reference pricing is used to determine the lowest ex-factory price of the medicine from France, Spain, Italy, Portugal, or Greece.
The price is converted to Turkish lira. A fixed rate of 60% of the previous year’s average exchange rate is now used for this calculation.
Mandatory discount rates are applied to determine the reimbursement price. For innovative medicines, this is often 41%.
Over the past few years, the above steps - particularly the sizeable mandatory discount and the use of fixed exchange rates that deviate significantly from the current exchange rate, have deterred some Pharma companies from launching innovative medicines in Türkiye.
Additionally, in November 2021 the Turkish Pharmacists' Association (TEB) reported drug shortages for 645 medicines, blaming the “significant difference” between the euro rate used in pricing calculations and the actual euro rate for the chronic supply issues.
(For more detail on the pricing methodology including statutory markups and internal reference pricing, I recommend the following resource.)
If Pharma is not willing to accept such price concessions – or the SSI is not willing to reimburse a new medicine, the good news is that there are other options available to Big Pharma such as the Alternative Reimbursement System (ARS). The ARS allows pharmaceutical firms to negotiate an alternative reimbursement model with the SSI if their product is not placed on the SSI reimbursement list.
This is essentially the route Pharma needs to take to implement a managed entry or risk-sharing agreement in Türkiye. It’s also a reimbursement pathway commonly used for medicines indicated for the treatment of rare diseases. To date, most agreements implemented have been finance-based agreements, not performance-based agreements that leverage agreed clinical milestones to determine final net pricing.
Limited willingness to reimburse ATMPs
Thus far the SSI has been unwilling to reimburse gene therapies for several reasons:
Their high cost makes funding them challenging for the SSI budget.
Limited long-term evidence leaves uncertainty around their cost-benefit analysis and long-term efficacy.
Lack of qualified trained staff and appropriate infrastructure to administer these treatments.
The availability of lower-cost alternative treatment options in some therapy areas.
The potential for greater healthcare access inequality.
In a few cases, patients in Türkiye have successfully gained access to gene therapy treatment abroad (such as in the USA or GCC) by leveraging crowdfunding to finance travel and treatment, but this solution is very costly - not just because of the typical list prices of gene therapies, but also because of the runaway inflation in Türkiye.
Are there any ways to bypass some of these pricing and reimbursement challenges?
Yes! In addition to the Alternative Reimbursement System mentioned earlier there’s also the named-patient use access route. It permits Turkish citizens to access medicines not approved by the Turkish MoH or that are not commercially available in exceptional cases.
How does it work?
At a physician's request, the Turkish Pharmacists' Association (TEB) is empowered to source medicines from overseas for individual patient use if:
There is an unmet medical need.
The medicine has received regulatory approval from either the EMA or FDA.
There is no marketing authorisation in Türkiye or if the product is otherwise unavailable there.
The process is quick and straightforward, plus it offers several advantages; full reimbursement is available at prices exempt from both international reference pricing and mandatory discounts, and the current exchange rate applies.
This access route has been used for multiple innovative drugs indicated for rare diseases and has been a key supply route for many high-cost, low-volume medicines.
Conclusion
Türkiye represents a key market in the CEE region but there are challenges for foreign innovative drug manufacturers to consider in their market access and pricing strategy.
Pharma needs to consider multiple criteria such as their product’s indication, the availability of alternative treatments, target pricing, international reference prices, and anticipated volumes in Türkiye to determine what is the most appropriate reimbursement pathway for their product.
Cover photo credit: Photo by Michael Jerrard on Unsplash