What is EUnetHTA and what’s happening next?
The HTA landscape in Europe is undergoing a transformation, and with that change comes new processes and new terminology – let’s explore!
The world of MA&P is full of acronyms and abbreviations (see what I did there?)
I suppose we must secretly love them or they would have run out of fashion by now.
One area with more acronyms and abbreviations than I can keep track of is EUnetHTA.
Alas, I bring you a simple guide on what and who EUnetHTA is and what’s coming next.
Are you ready for acronym and abbreviation overload?
Here goes…
HTA
Meaning = Health Technology Assessment
What is it?
The systematic evaluation of the properties, effects, and/or impacts of a health technology. Its purpose is to inform health policy and reimbursement decision-making for health technologies. HTAs typically assess the clinical effectiveness (medical), cost-effectiveness (economic), ethical, and social aspects of a health technology.
EUnetHTA
Meaning = European Network for Health Technology Assessment
What is it?
EUnetHTA began as an informal project-based collaboration between several European HTA agencies back in 2005. Since then, it’s grown into a broader HTA network, whose work paved the way for the implementation of the HTA Regulation (see below).
EUnetHTA’s primary goal is to create an effective HTA network and framework in Europe that develops reliable, timely, transparent, and transferable information or advice on innovative medicines, medical devices, and procedures.
By pooling resources and expertise across Europe, EUnetHTA’s processes and methodologies seek to ensure efficient use of resources, while strengthening the quality of HTA across Europe.
EUnetHTA’s work includes:
Developing HTA tools, methodologies, models, and guidelines
Implementing joint work on assessments
Promoting good practices
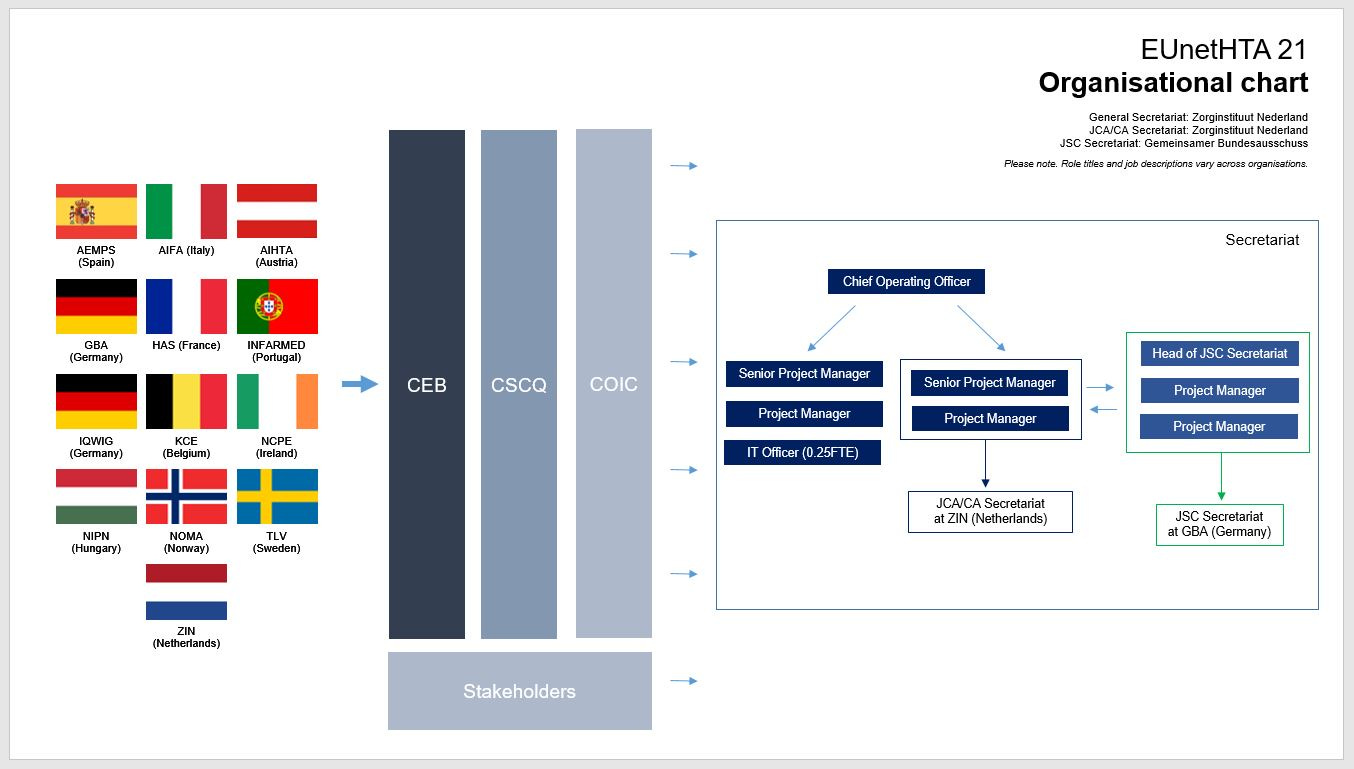
EUnetHTA is structured as a Secretariat, an Executive Committee, a Stakeholder Forum, and a range of topic-specific Working Groups and Work Packages. The full list of member organisations of EUnetHTA can be found here.
It’s important to note that although the name EUnetHTA is most commonly used, it has had several slightly different titles over the last couple of decades. Each title links to a distinct time period and a specific set of objectives and activities.
Note: JA = Joint Action.
EUnetHTA 21
Meaning = as shown above, you can think of it as EUnetHTA version 6.0, only when renaming, the year of implementation (2021) was selected rather than the version number.
What is it?
The EUnetHTA 21 joint consortium* took over from EUnetHTA and will run from 2021 to 2025, after which it will be replaced by the HTA regulation. Its current activities include conducting Joint Clinical Assessments of medicines, developing methodological guidelines, and implementing capacity-building training.
*The consortium is led by ZIN (The Netherlands) and includes 44 partner organizations from 28 countries, primarily HTA bodies from across Europe.
Note: The image below is sourced from and the property of EUnetHTA.
HTA Regulation
Official title = Regulation (EU) 2021/2282 on Health Technology Assessment. See more here.
What is it?
The HTA Regulation will introduce a permanent EU HTA system and replace EUnetHTA from January 2025.
It requires EU countries to participate in joint scientific consultations and clinical assessments following established processes developed by the EUnetHTA 21 consortium.
Its implementation and governance will be overseen by the HTA Coordination Group of national authorities and the European Commission.
JSC
Meaning = Joint Scientific Consultations
What is it?
JSCs are a mechanism for early dialogue between health technology developers and EUnetHTA partners. Discussions will focus on a health technology’s development plans and/or evidence requirements. They seek to provide developers with feedback and guidance to optimize data collection through reviews of clinical study design, patient populations, comparators, and outcomes.
The output of the process is a consensus document that summarizes the scientific advice from participating HTA bodies.
JCA
Meaning = Joint Clinical Assessments
What is it?
JCAs are collaborative assessments of the relative effectiveness of health technologies. Currently, they are jointly conducted by multiple European HTA organizations through EUnetHTA.
JCAs seek to jointly assess the evidence on a technology's clinical effectiveness and safety compared to standard care, and in doing so create a single joint assessment that can be used by multiple European countries to avoid duplication of work.
Ultimately, the JCA should help inform national/local HTA processes and policy decisions on the adoption, pricing, and reimbursement of new health technologies.
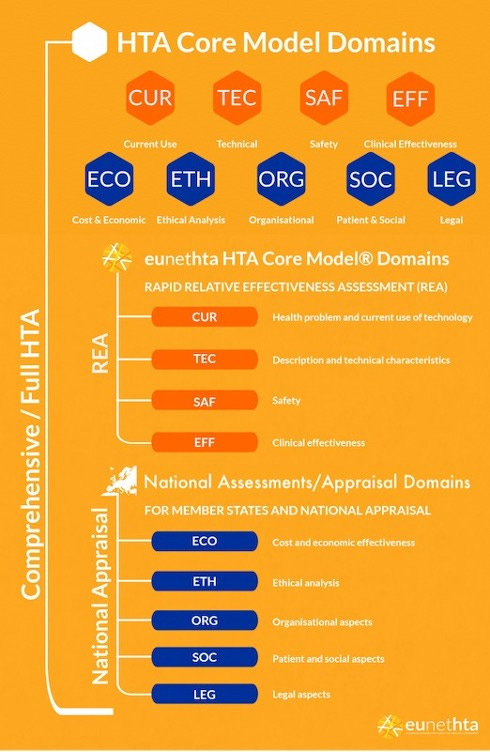
HTA Core Model®
Meaning = …yes, I cheated…this one isn’t an abbreviation, but it contains many of them hence I’ve included it. See EUnetHTA’s HTA Core Model® diagram below!
What is it?
The HTA Core Model® is a methodological framework developed for the production and sharing of HTA information. It has nine domains, all of which are used in a full HTA assessment.
Note: The diagram is sourced from and the property of EUnetHTA.
REA
Meaning = Rapid Relative Effectiveness Assessments
What is it?
Pretty much what it says on the tin – REAs are streamlined collaborative assessments that evaluate the relative effectiveness of health technologies. Unlike full HTA assessments, they will only cover the four domains of the HTA Core Model® and do not include any economic analyses or systematic literature reviews.
The outputs of REAs will be available in a timelier manner (approx. 2-4 months) to inform pricing and reimbursement decision-making.
Wrap up
There are other acronyms and abbreviations associated with EUnetHTA but these are the key ones you need to know in order to follow along with what’s happening at a high level.
For more information, you can review the EUnetHTA website, which has a helpful overview video here.
If you found this blog post helpful, please consider sharing it or subscribing :)
Cover photo credit: Photo by Alina Grubnyak on Unsplash