Lessons from recent EU payer decisions on gene therapies
Last Monday was another landmark day for gene therapies as a new and potentially curative one-off gene therapy for babies with spinal muscular atrophy (SMA) looks set to become the most expensive treatment ever recommended by NICE.
So let’s take a closer look at the lessons that can be learned so far from recent EU payer decisions on gene therapies.
The overarching lesson:
EU payer experience with gene therapies is limited but has highlighted the challenges of applying conventional Health Technology Assessments (HTA) methods and reimbursement strategies to high cost, potentially curative therapies.
Limited Evidence Packages
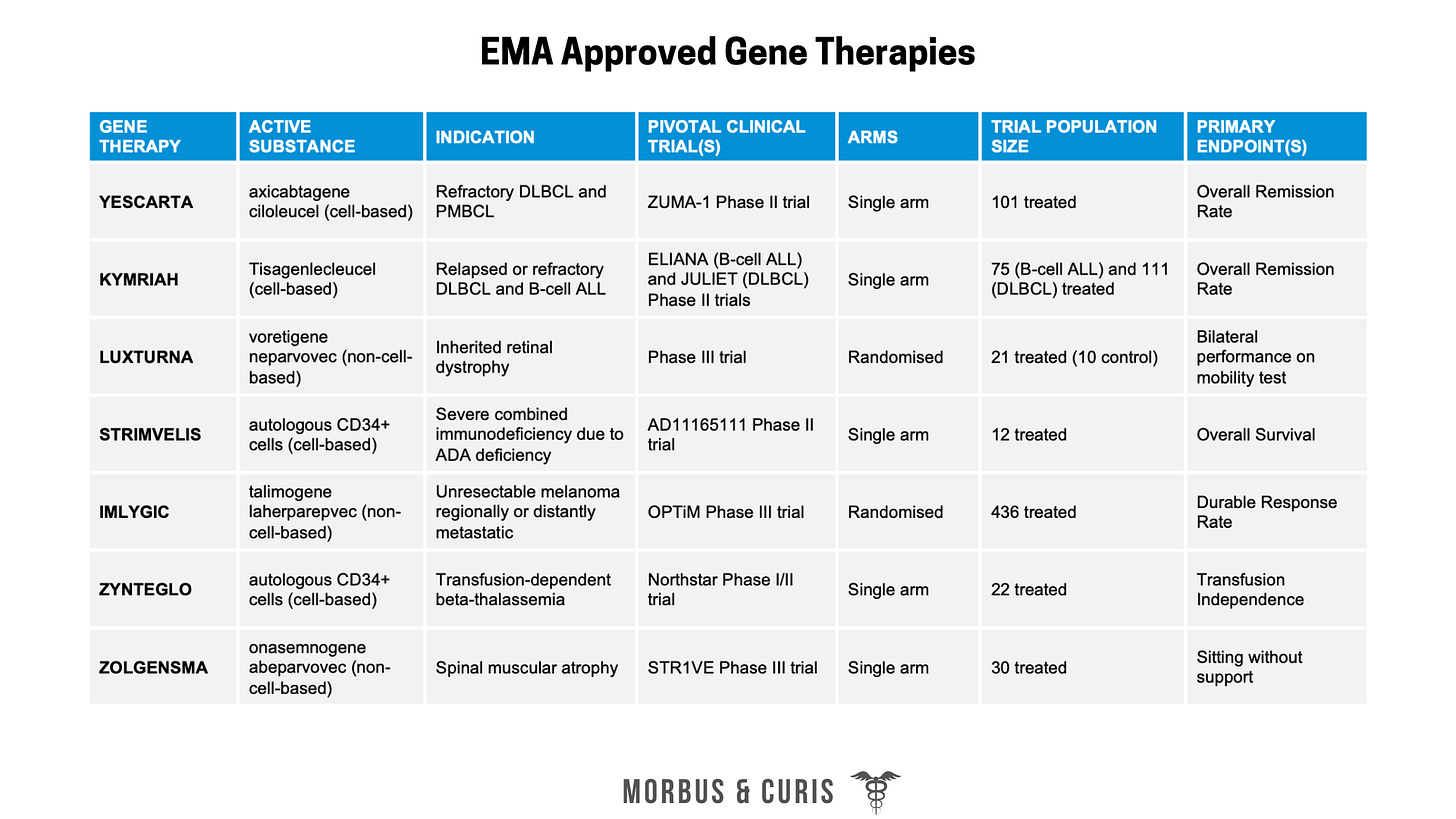
The table below outlines gene therapies that currently have EMA Marketing Authorisation in Europe.
Source for information.
One of the first things you might notice is that the list is not long!
Another key takeaway from the information is that:
Gene therapies often attain regulatory approval with limited evidence packages.
Take a closer look at the table and you’ll notice several common themes that contribute to the limited evidence package of gene therapies at launch:
Some were approved via PRIME* resulting in accelerated regulatory approval, which can limit the evidence base for a treatment that demonstrate high efficacy in early clinical trials (*PRIME = when a product is designated breakthrough status, its developer qualifies for enhanced interaction with the EMA to optimise the product’s clinical development plan and speed up its evaluation, which facilitates accelerated regulatory approval).
Many gene therapies treat rare diseases and consequently have low patient numbers enrolled in their clinical trials since larger recruitment can be prohibitively expensive and time-consuming.
Clinical trials for gene therapies often lack a comparator arm. This lack of comparative clinical data creates challenges for payers that need to quantify a treatment’s degree of added clinical benefit versus the standard of care. In the extreme, this can result in a dossier being deemed incomplete or rejected.
The serious and progressive symptoms that characterise many genetic disorders raise ethical and practical barriers to conducting randomised clinical trials (RCTs) with a comparator (active or placebo) arm. Consequently, single-arm trials or RCTs with early cross-over are likely to be common standards for regulatory approval. An added complication is that many rare diseases lack a standard of care treatment so it can be challenging to determine an appropriate comparator.
Some gene therapy trials have used surrogate endpoints. This can occur because some rare diseases lack a validated and standardised patient health outcome measure.
To summarise - the novel characteristics of gene therapies (i.e. their complex and often personalised nature) and the fact that many treat rare diseases mean that it is challenging for gene therapy manufacturers to generate the robust clinical evidence package expected by EU payers.
Conditional Reimbursement
Pricing and reimbursement (P&R) decision-making follows regulatory approval and recent P&R decisions for gene therapies have demonstrated that:
Many EU payers are only willing to approve conditional reimbursement of gene therapies at launch.
The limited evidence package of gene therapies at launch, coupled with their high price tags, creates a high degree of uncertainty around their clinical and cost-effectiveness.
As a result, EU payers are opting to manage the risks of reimbursing high-cost gene therapies, with limited clinical evidence packages, via managed entry agreements (MEAs).
These agreements range from temporary approval of a therapy for a fixed time period while further evidence is gathered before a future P&R reassessment, to others that are pay-for-performance contracts, linking payments or rebates to defined clinical milestones measured at agreed time points.
While the CAR-T cell therapies, Yescarta and Kymriah, were able to attain reimbursement in all EU5 countries, many gene therapies have failed to do the same, highlighting the strict evidence requirements and cost-constraints being managed by some EU payers.
Table completed using publicly available sources. The specific details of many managed entry agreements and contracts are confidential.
High Pricing
I’ve already called gene therapies high-cost, which is the next lesson:
Gene therapy list pricing is very high and highly consistent across the EU5 markets.
The below graph shows some of the publicly available list prices (proposed or approved) of gene therapies across the EU5.
It’s worth noting that these list prices only pertain to the gene therapy product, and none of the associated costs of treatment (i.e. apheresis, administration, hospitalisation and managing toxicities) and because of the complex nature of gene therapies, these costs should not be underestimated!
Payers in France are taking steps to address these financial challenges and recently implemented a specific funding code for Kymriah and Yescarta that provides authorised hospitals with €15,000 to help cover the costs associated with patient hospitalisation during treatment.
Graph completed using publicly available sources. The specific details of negotiated contracts (e.g. discounts and rebates) are confidential.
How are these prices justified? Gene therapy manufacturers have so far defended their prices by pointing to high R&D and manufacturing costs, the small patient population from which they can recover R&D costs and the transformational, potentially curative therapeutic effectiveness of gene therapies.
If longer-term evidence demonstrates that some one-off gene therapies are curative, then manufacturers can argue that they significantly reduce the total healthcare bill of a patient by reducing or eliminating certain long term care costs (i.e. chronic treatments and management of comorbidities).
However, these price tags pose severe affordability challenges to healthcare systems that are not set up for very high, one-off payments.
Funding Flows
An interesting observation from Europe is how varied funding of gene therapies can be. A key takeaway from the different funding flows is an unsurprising one; centralised, supplementary funding is less likely to create provider level price-related access hurdles.
Below is an overview of the funding flows and data collection for Yescarta in the EU5.
Data collection is particularly important for gene therapy funding because it’s linked to payments/rebates and/or will inform future P&R reassessments.
Health Technology Assessments
HTAs are used to determine a reasonable price range that is aligned to the degree of clinical benefit demonstrated. Recent HTA decisions for gene therapies have highlighted the next lesson:
Most HTA frameworks do not make special allowances for gene therapies, therefore payers apply the same criteria and methodologies used for conventional medicines.
Below are some of the challenges faced:
If EU payers wanted to make special allowances for gene therapies, there are multiple ways this could be achieved. Two potential solutions include:
Provide greater flexibility or more lenient thresholds for gene therapies approved via existing HTA frameworks
Create a new P&R approval route that gene therapies are eligible for - this could be something analogous to NICE’s HST pathway - or an Advanced Therapy Medicinal Product (ATMP)-specific HTA pathway that could provide greater clarity and flexibility on acceptable data packages, cost-effectiveness thresholds and reimbursement procedures.
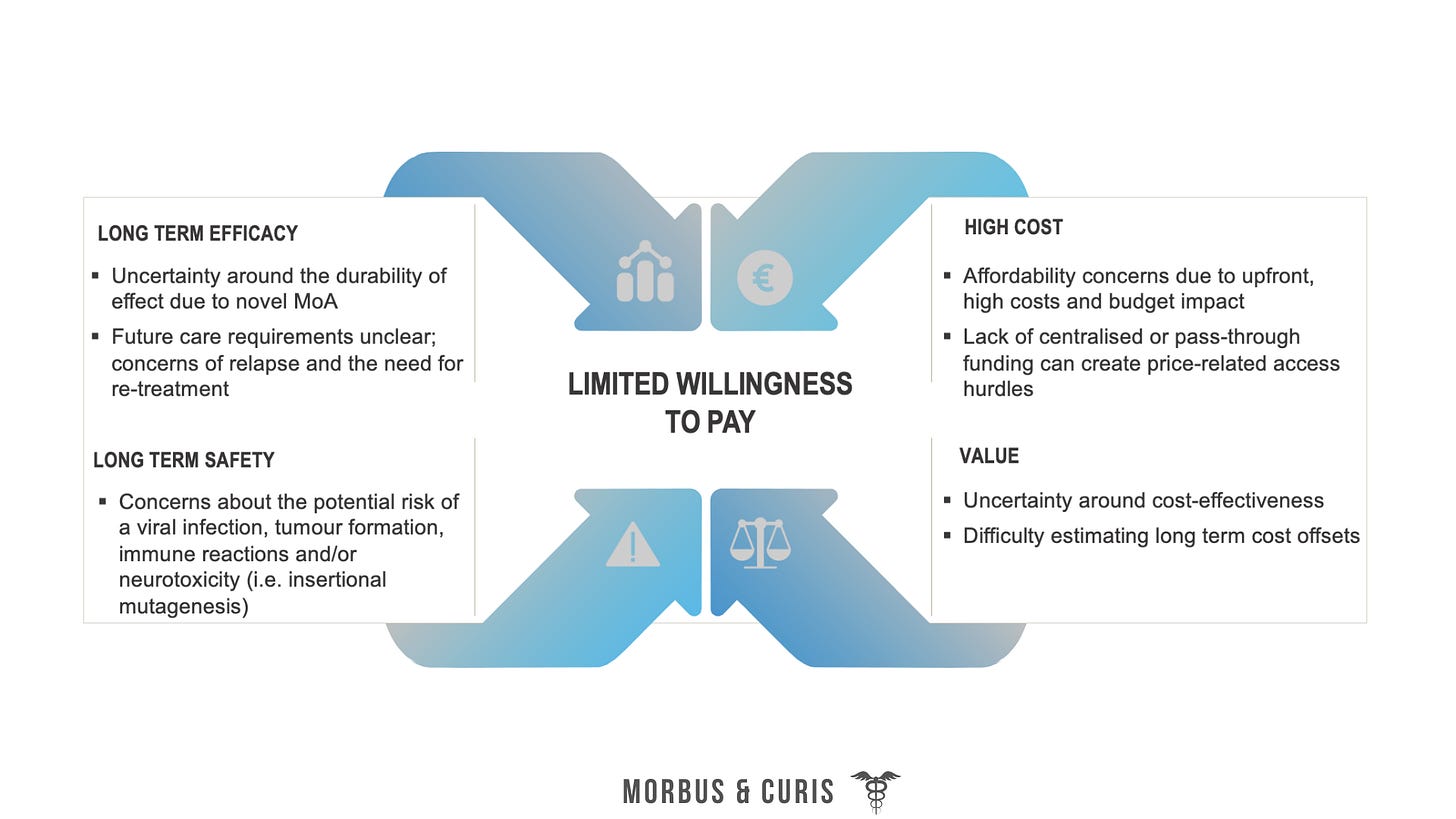
Value Perceptions and Willingness to Pay
While payers recognise the innovativeness of gene therapies, their high costs and limited evidence packages have restricted perceptions on value and willingness to pay.
In spite of these uncertainties, some payers have been willing to conditionally approve reimbursement.
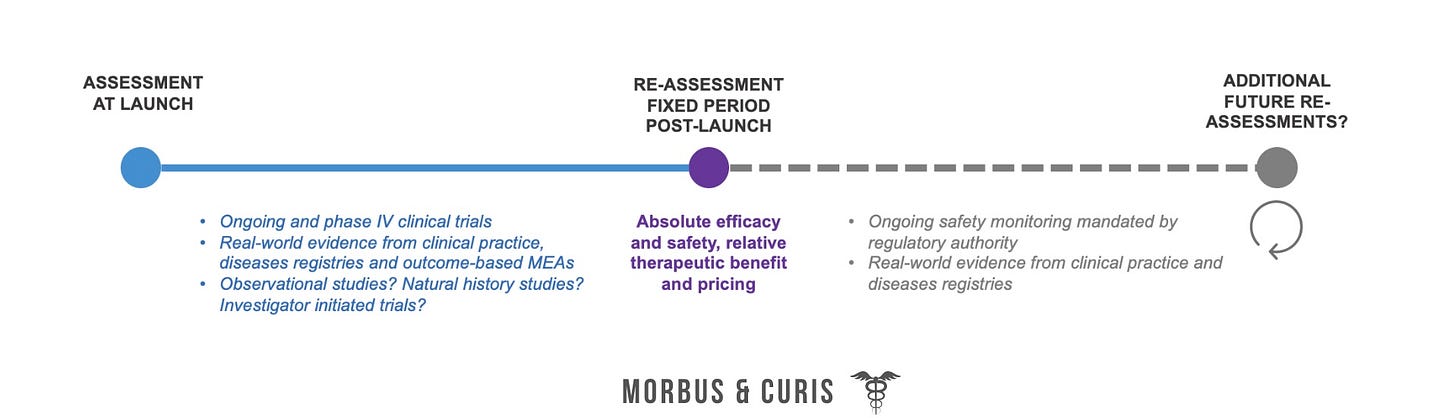
Further Evidence Generation and Reassessments
Many EU payers have mandated further evidence generation and future reassessments of gene therapies to mitigate uncertainties around long term efficacy and safety and value.
Payers, in addition to regulatory authorities, are mandating patient monitoring and data collection
Some payers are mandating the implementation of disease registries and/or specific data collection requirements (Germany is a good example of this emerging trend).
The industry could partner with payers to understand which study designs would best address uncertainties.
Future reassessments will seek to mitigate uncertainties regarding the long-term efficacy and safety and pricing of gene therapies.
Innovative Payment Models
Innovative payment models are becoming more widely accepted and adopted by EU payers to control the financial challenges of high-cost gene therapies with limited long term data.
Not all health service funding infrastructures are fully optimised for high-cost gene therapies. Payers managing fixed yearly or local budgets, in particular, may be less willing to pay for gene therapies due to affordability challenges. Potential solutions to affordability challenges include:
Funding gene therapies via pass-through funding, separate from DRG tariffs and hospital budgets.
Using annuity payments to spread the large upfront costs of gene therapies over a wider period of time.
Negotiating pay-for-performance contracts, which can reduce the budget impact and financial risks of gene therapies by linking payments or rebates to patient outcomes. In order to be implemented successfully, these contracts require manufacturer and payer alignment on simple measurable definitions of treatment success and failure.
EU payers are also utilising other approaches to contain the budget impact of high-cost therapies such as affordability thresholds, volume caps or volume agreements, restrictions on patient populations, special funding for high-cost drugs and limits on pharmaceutical spending growth.
Conclusions
Payer experience with gene therapies is limited but has highlighted multiple market access and pricing challenges.
Firstly, payers are willing to deem gene therapy evidence packages as insufficient or invalid despite the challenges of generating robust data for small patient populations and/or personalised treatments.
Most EU HTAs bodies are unwilling to treat gene therapies differently versus conventional medicines.
Long term and real-world clinical uncertainties, as well as high prices, are challenging for payers to accept.
Early payer engagement to align on the clinical development programme could improve payer acceptance of a gene therapy’s evidence package at launch.
Secondly, conditional reimbursement approaches are being used to address uncertainties.
Payers are more willing to negotiate MEAs for gene therapies versus conventional medicines, and they have been a common success factor for reimbursement and pricing approval.
MEAs help address clinical uncertainties through evidence generation and can mitigate some financial challenges faced by payers.
Thirdly, some EU funding frameworks are not yet optimised for gene therapies.
Hospital budgets and existing DRGs tariffs cannot cover high-cost gene therapies and their associated costs without supplementary funding.
Centralised, pass-through funding or new DRGs could help remove provider level affordability barriers.
It is unclear how some gene therapies will be funded once temporary funds are finished or competition for certain funds increases.
Finally, recent events and manufacturing advancements may require some of these lessons to be revisited.
For example:
The COVID-19 pandemic is likely to result in new austerity measures or cost containment measures across Europe. These could put pressure on healthcare budgets and high-cost therapy prescribing.
Brexit has changed the regulatory landscape for gene therapies in Great Britain, which could impact market access to gene therapies. Whether that impact is positive or negative remains to be seen.
Advancements in the quality and scale of viral vector production could significantly reduce the manufacturing costs associated with gene therapies. This could help payers to negotiate lower prices for future gene therapies.
If you found this blog post helpful, consider sharing it or subscribing :)